Polyester
One atom away from nylon, with the working-class job description.
- Volume
- 01
- Order
- № 03
- Read
- 13 min
- Published
Nylon is the prestige fiber of the bag world. Polyester is the working one. It shows up everywhere (lining fabrics, gym bags, X-Pac face sheets, the films inside DCF) and gets credit for very little of it. The chemistry is one bond away from nylon and the practical differences are bigger than that suggests.
The ester bond, in one paragraph
Nylon chains hold each other through hydrogen bonds at every amide group (–C(=O)–NH–). The nitrogen-attached hydrogen reaches across to a neighboring chain’s oxygen and clings. Polyester replaces that nitrogen with another oxygen: an ester bond (–C(=O)–O–). One atom different. The new bond cannot offer an N–H to its neighbors, so inter-chain grip drops.
The two bond geometries:
- Amide:
–CO–NH– - Ester:
–CO–O–
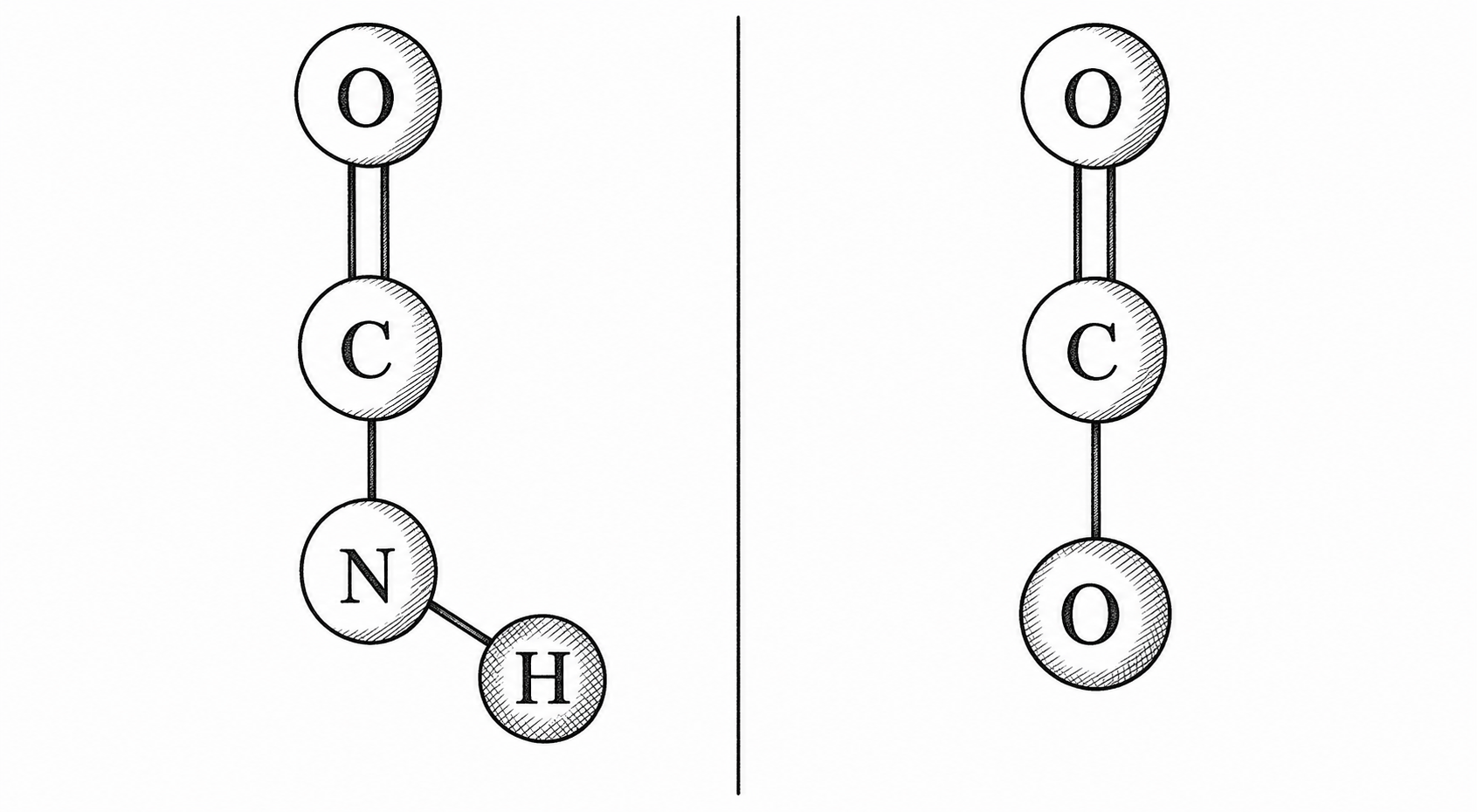
One atom difference. Polyester’s oxygen where nylon has nitrogen means less inter-chain grip but better UV and moisture performance. Illustrative only.
That single substitution (nitrogen replaced by oxygen) changes the material’s character significantly. Oxygen cannot donate hydrogen bonds the way nitrogen can. Ester chains are slicker; they do not grip each other as tightly. The result: polyester is somewhat less strong than nylon at equivalent denier, less elastic, and a bit more rigid.
But it is not a worse bond, just a different one. Ester bonds have their own advantages, which we will get to.
What is polyester?
The full chemical name is poly(ethylene terephthalate), PET. You already know PET: it is the plastic in water bottles, food containers, blister packs, and the Mylar films in X-Pac and DCF (chapters 3 and 5).
The two building blocks are:
- Terephthalic acid (a ring-shaped aromatic molecule with two acid groups)
- Ethylene glycol (a simple two-carbon diol, basically antifreeze)
These react via condensation polymerization called esterification: each acid group reacts with a hydroxyl group, water is driven off, and an ester linkage forms. The chain grows until you have long-chain PET with molecular weights in the 20,000–40,000 g/mol range for fiber-grade material.
Polyester is the most widely produced synthetic fiber in the world, surpassing nylon by a wide margin. Made from oil-derived feedstocks (paraxylene → terephthalic acid; ethylene → ethylene glycol), so it shares nylon’s petrochemical origin.
Making fiber: melt spinning (again)
Like nylon 6,6, polyester is melt-spun. PET melts at around 260°C (vs nylon 6,6’s 265°C, nearly identical), is pushed through a spinneret, cooled in air, and mechanically drawn.
The drawing step matters. When PET exits the spinneret, the molecular chains are still randomly oriented. Drawing (mechanically stretching the fiber to 3–5× its original length) forces chains to align along the fiber axis, increasing crystallinity and tensile strength dramatically. PET responds slightly differently than nylon: because the benzene ring in terephthalic acid is geometrically rigid, PET chains pack into crystalline regions more readily. More crystallinity means better shape retention and lower elasticity, which is why polyester bags keep their shape better but recover less well from impacts.
Density: the number that changes everything
Here is where polyester diverges sharply from nylon in ways that matter for bags.
- Nylon 6,6 density: 1.14 g/cm³
- Polyester (PET) density: 1.38 g/cm³
Polyester is 21% denser than nylon. Not a small difference. A 500D polyester yarn contains the same mass per 9,000 meters as a 500D nylon yarn (that is what denier means), but since polyester is denser, those 9,000 meters are thinner than nylon at the same denier.
The practical implication: at the same denier rating, a polyester fabric is slightly thinner and less voluminous than the equivalent nylon fabric. Polyester packs tighter into a weave. Bag manufacturers can hit a higher thread count with polyester at the same denier, partially compensating for its lower strength-to-weight ratio with a denser weave.
This also explains the X-Pac numbers: the RX-series and RVX-series use polyester face fabrics, and they are slightly heavier per square meter than the equivalent denier nylon variants, because a denser base fiber = more mass in the finished fabric.
The four key differences from nylon
Polyester and nylon are often treated as interchangeable in bag discussions. They are not. Here are the structural reasons they perform differently.
1. Water absorption: polyester wins decisively
Nylon 6 absorbs ~3.5–4% of its weight in moisture; nylon 6,6 absorbs 2.5–3%. Absorbed water plasticizes the nylon chains, reducing strength by ~10–15% when saturated, stretching the fiber slightly, and significantly increasing dry time.
Polyester absorbs less than 0.5% moisture. Essentially zero. Water does not penetrate into the fiber structure. A polyester bag does not get measurably heavier when wet, does not stretch in humid climates or contract in dry climates, and dries rapidly because there is no absorbed water to drive off, only surface moisture.
Not a minor practical advantage for outdoor bags. A nylon bag carried in Southeast Asia humidity can absorb enough moisture to noticeably affect drape and fit. Polyester is dimensionally stable across humidity ranges.
2. UV resistance: polyester wins decisively
Nylon degrades under UV light fairly readily. Nylon’s amide bonds do not absorb UV energy well, but the resulting degradation still leads to chain scission, fading, and embrittlement over years of outdoor exposure.
Polyester’s ester bond undergoes UV degradation differently. PET does degrade under UV (Norrish-type chain scission at the ester carbonyl), but significantly more slowly than nylon under the same conditions, and the benzene ring in PET actually absorbs some UV energy, acting as an internal light stabilizer. Practically: an untreated polyester bag left in a sunny car for years holds its color and structure substantially better than an untreated nylon bag.
This is why polyester dominates in applications that require long outdoor UV exposure: awnings, outdoor furniture fabric, marine covers.
3. Strength-to-weight ratio: nylon wins
Nylon has a higher tensile strength-to-weight ratio than polyester. At the same denier, nylon fabric is generally stronger in tensile and tear tests. Polyester can partially compensate by using denser weaves, but a well-constructed polyester fabric still typically falls short of equivalent nylon in raw mechanical strength.
This is the main reason premium, structural, heavy-use bags default to nylon: panels facing constant load-bearing stress, shoulder attachment points, and bottom panels all benefit from nylon’s higher tensile strength.
4. Odor retention: nylon wins (by a lot)
Uncomfortable for polyester. Research published in Applied and Environmental Microbiology found that polyester garments harbor significantly higher populations of Micrococcus bacteria (a genus strongly associated with unpleasant odors) than cotton equivalents. A University of Alberta study found that odorant compounds (from sweat) are more attracted to polyester than to other fibers, and do not fully wash out.
The mechanism: polyester is hydrophobic, so sweat stays on the fiber surface rather than being absorbed and wicked away. Bacteria that break down fatty acids into smelly volatile organic compounds (particularly Micrococcus) adhere strongly to polyester’s smooth, non-polar surface and form biofilms that resist regular washing. Over repeated use, VOCs accumulate.
This matters for bag linings and straps more than face fabric. If you have ever noticed an older polyester-lined bag smelling persistently stale despite washing, this is why.
Weaves: same as nylon
Polyester uses the same weave families as nylon: plain weave, basket weave (ballistic-style), and ripstop. Same geometric principles apply. Same denier-to-weight math (adjusted for density). Same PU and DWR coatings on the back and face.
One difference worth noting: polyester takes color more readily than nylon. Disperse dyes bond effectively with PET’s crystalline structure, producing vibrant, light-fast colors that resist UV fading for years. Nylon can also be dyed, but colors fade faster under UV. Another reason brands that prioritize consistent color across a product line lean toward polyester.
Cordura polyester: yes, it exists
When most bag nerds hear “Cordura,” they picture nylon. But Cordura is a brand, and Cordura makes polyester fabrics too. Cordura polyester comes in similar denier ranges (600D, 900D, 1000D) with the same basket weave structure as Cordura nylon ballistic.
Who uses it? JanSport is one of the most cited examples: the brand transitioned from nylon to polyester Cordura and now uses no nylon in current bags. Budget-range bags from mass-market brands almost universally use polyester rather than nylon, because polyester is cheaper. Not necessarily a performance indictment: for a casual school bag that lives under a desk, polyester’s UV stability and shape retention matter more than nylon’s superior abrasion resistance.
rPET: the recycled angle
This is where polyester has a genuine ecological narrative that nylon currently lacks at scale.
rPET is polyester fiber made from post-consumer plastic waste, predominantly PET bottles. The process: bottles are collected, labels removed, shredded into flakes, washed, melted, and re-extruded into fiber using the same melt-spinning process as virgin PET. The fiber is chemically identical to virgin PET.
Production of rPET requires approximately 50–75% less energy than virgin polyester and uses no new petroleum feedstock. This has driven broad adoption:
- Dimension-Polyant’s RX-series X-Pac uses 100% post-consumer recycled polyester face fabric and is certified cradle-to-gate climate neutral (Chapter 3).
- Patagonia, Cotopaxi, Osprey have integrated rPET broadly across product lines.
- Most promotional/tote bags are now rPET rather than virgin polyester.
The honest caveat: recycling is not circular. rPET fiber cannot easily be recycled again into fiber, it typically downcycles to industrial fill or landfill at end of life. True chemical recycling (breaking PET back to its monomers and repolymerizing) exists but is not yet industrial-scale. rPET is better than virgin, but “sustainable” is a stretch.
Where the ester bond is actually a feature
The ester bond is hydrolytically cleavable: it can be broken by water under acidic or basic conditions. For structural fiber, this seems like a weakness. And it is, mildly, for long-term degradation in very wet conditions. But this same property is what makes PET chemically recyclable: enzymatic and chemical PET recycling processes work precisely by hydrolyzing ester bonds back to terephthalic acid and ethylene glycol.
In laminates (X-Pac, DCF), thin PET films are processed in a way that maximizes crystallinity, which dramatically slows hydrolysis by limiting water diffusion into the crystalline regions. Those PET films are not going to hydrolyze on your bag under normal conditions. But the chemistry is the underlying reason that PET recycling is more technologically tractable than nylon recycling, a factor that will matter increasingly as sustainability requirements tighten.
TPU coating: the thermoplastic alternative to cast PU
Throughout this book the default waterproofing for woven fabrics has been a PU (polyurethane) coating: a polymer dissolved in solvent, painted onto the back of the textile, and cured. Effective and cheap, but consumable: PU coatings crack at fold lines, can hydrolyze in humid storage, and degrade unevenly over years. Chapter 1’s “Where it loses” section names this directly: the PU coating is the failure mode that defines the lifetime of a standard nylon bag.
TPU (thermoplastic polyurethane) is a different processing route to a closely related polymer. Instead of dissolving the urethane in solvent and brushing it on, TPU is melted and either extruded as a thin film or heat-laminated directly onto the substrate. Same urethane chemistry family; fundamentally different application.
What changes when the coating is heat-formed instead of solvent-cast:
- Film uniformity. Solvent-cast PU varies in thickness wherever the coating pools or thins. A heat-formed TPU layer is geometrically flat and reproducible across a roll.
- Adhesion. TPU bonds to polyester (and to itself, at seams) by partial fusion at the interface, not by mechanical keying into the textile’s surface texture. The bond is harder to peel and tolerates flex cycles better.
- Surface behavior. TPU presents a smoother, less porous outer face. Grime sits on top instead of working into a textured coating. The “wipes clean” reviews of TPU-finished bags are real, not cosmetic.
- Hydrolysis resistance. TPU formulations are typically more hydrolysis-resistant than the polyester-based PU coatings most often used on bag interiors, which is why TPU shows up on rain shells, dry bags, and waterproof zipper tapes where standard PU would fail in months.
The honest framing: TPU coating is a cheaper, non-structural analog to the PET film in X-Pac. Both put a thermoplastic film between water and the bag’s contents. X-Pac (Chapter 3) bonds that film inside a four-layer laminate with a bias-grid scrim: structural, expensive, dimensionally stable under sail-grade load. A TPU-coated polyester puts the film on the back of a single woven layer: waterproof and durable, but with no scrim, no laminate stack, and none of the $40/yard fabric cost. Two different solutions to the same problem at very different price points. For most daily-carry use cases (laptop in, laptop out, occasional rain, occasional spill), the simpler TPU-coated weave does what the laminate would do, at a fraction of the price.
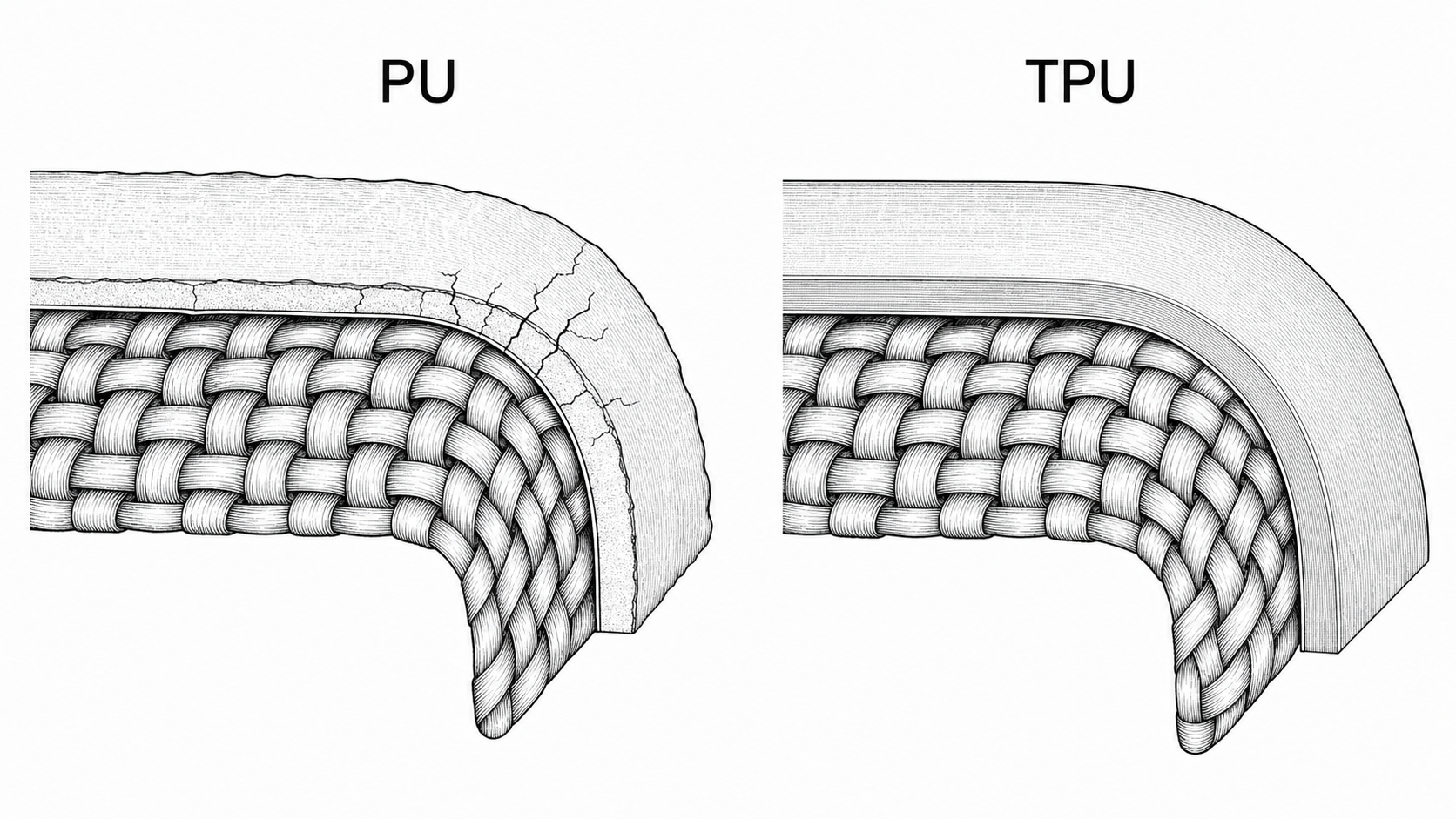
PU and TPU both waterproof by coating the textile, but TPU behaves more like a thermoplastic film: tougher, more uniform, easier to wipe clean. Illustrative only.
Polyester in the Inventory
The Everest gym bag uses 600D polyester, 32L. That is appropriate: a gym bag needs to be UV-stable, wipe-clean, dimensionally consistent in a humid environment, and cheap enough to not care about. 600D polyester does all of that without pretense.
 Everest 32L, 600D polyester, separate wet-pocket end compartment. UV-stable, wipe-clean, cheap. Illustrative only.
Everest 32L, 600D polyester, separate wet-pocket end compartment. UV-stable, wipe-clean, cheap. Illustrative only.
Polyester is also the face fabric in EcoPak (Chapter 4), and the film layers in both X-Pac (Chapter 3) and DCF (Chapter 5) are PET: polyester in a different form.
Lineage backpack: what polyester taught about integrated tech
Solgaard Lifepack. Shore-Tex shell: proprietary 100% recycled-PET ripstop, in the rPET family discussed above. Integrates with the Solarbank: 15,600 mAh, IPX7-rated, automatic solar charging via a removable PV panel, 4–5 iPhone full charges per cycle. The bag itself is straightforward 1680D-equivalent rPET; the differentiation is the energy system. What this taught: integrated tech in a bag is a category trap. The solar panel charges slowly and only when oriented correctly. The battery is heavier than carrying a standalone power bank that lives in a pouch. Useful as a single object on a long flight; awkward as daily carry. The lesson is not about Shore-Tex (which performed exactly as rPET should, UV-stable, wipe-clean, dimensionally honest) but about systems vs objects. A bag is better when it does one thing well; charging belongs in a Pioneer-style global pouch that moves between bags, not soldered into a shell. The fabric was right. The integration was a detour. (solgaard.co)

Solgaard Lifepack, Shore-Tex (100% recycled PET) ballistic shell, recessed dark solar window upper-left feeds Solarbank battery. Illustrative only.
Travel and overnight backpack: what TPU-coated polyester taught
Portland Gear Cascade Standard 21L (Navy). Acquired August 2025. Used in the household for travel and overnight trips, the only TPU-coated polyester in the collection, and the construction is exactly what the previous subsection describes. 100% polyester face fabric with TPU matte coating: one woven layer, TPU on the back doing the waterproofing, no laminate stack, no bias scrim. 16” H × 11” W × 8.5” D, 2.375 lbs (38 oz), 16” laptop sleeve. Water-sealed zippers, laser-perforated bottle holsters, wireframe pop-top, luggage sleeve, hidden security back pocket. What this taught: polyester is not the lesser fiber, it is the working one. This entire chapter sits between Ultra 400X (UHMWPE laminate, premium tier) and EcoPak (rPET laminate, mid tier), and the Cascade does the same travel-and-overnight job at the value tier with 100% polyester and a TPU finish. The pop-top wireframe is the design move that earns it: instead of fighting polyester’s higher rigidity (the Ch.2 chemistry: more crystalline than nylon, holds shape but does not drape), the wireframe makes the stiffness structural: the bag stands open, the opening stays where you put it, the coating and frame work as one part. The navy colorway is the disperse-dye discussion above made visible: PET takes disperse dyes into the crystalline regions, the color reads as a deliberate finish rather than a budget concession, and it will stay that way for years of UV exposure. The lesson is not “buy the cheap option.” The lesson is the right material for the role. A travel and overnight pack that goes through hotel floors, airline overheads, and the occasional wet seat does not need a bonded laminate: it needs a smooth, wipeable, dimensionally stable shell that holds shape and packs flat against a luggage sleeve, which is exactly what TPU-coated polyester delivers. (stilessupply.com, apartmenttherapy.com, esquire.com)

Portland Gear Cascade Standard 21L, 100% polyester with TPU matte coating, wireframe pop-top, navy. Illustrative only.
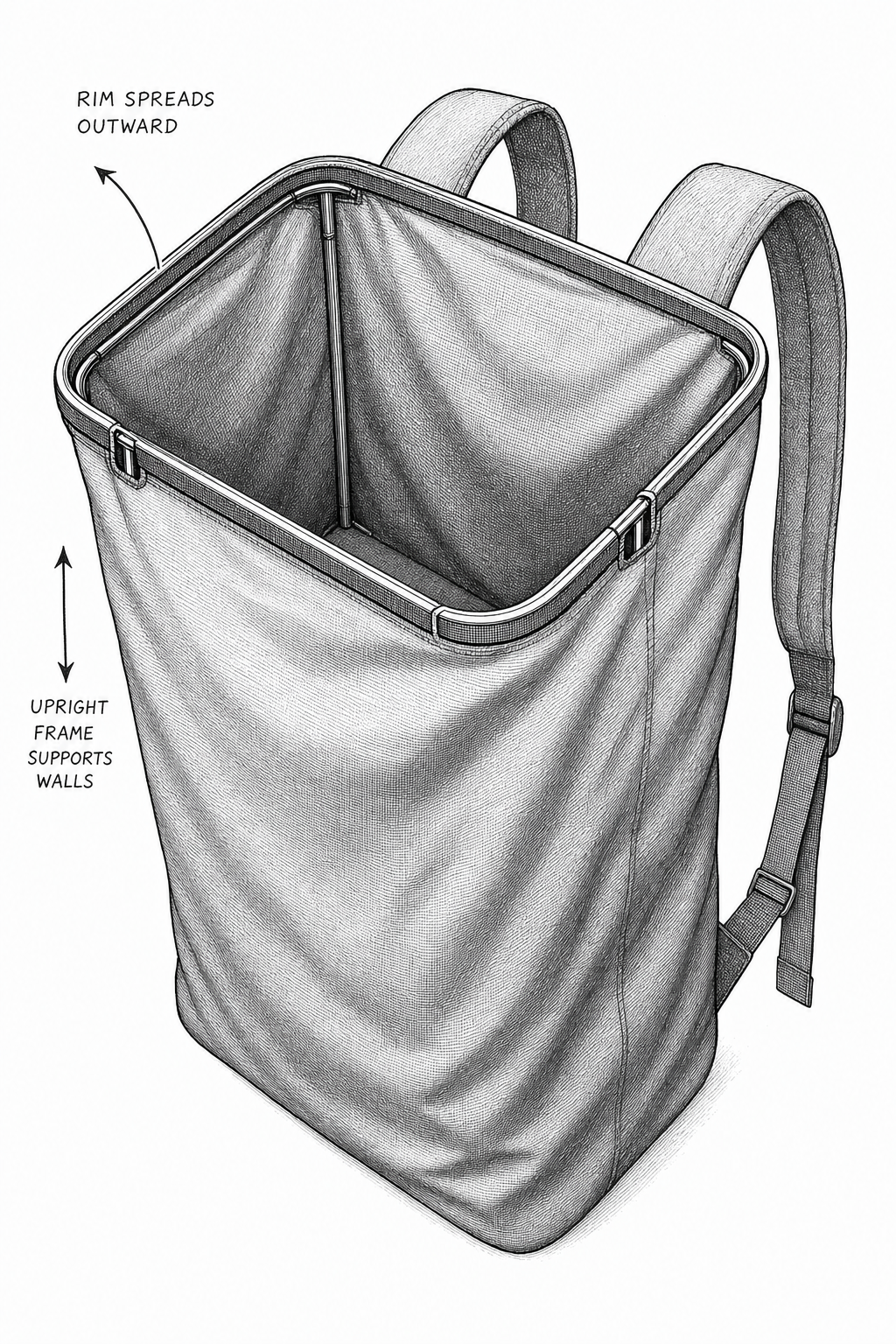
The Cascade’s wireframe pop-top turns a soft coated polyester shell into a self-presenting opening. Structure comes from hardware plus fabric stiffness, not fabric alone. Illustrative only.
The Master Comparison (so far)
| Property | Nylon 6,6 | Polyester (PET) |
|---|---|---|
| Base bond | Amide | Ester |
| Density (g/cm³) | 1.14 | 1.38 |
| Moisture absorption | ~3–4% | <0.5% |
| UV resistance | Moderate | Excellent |
| Strength-to-weight | High | Moderate |
| Color retention | Good | Excellent |
| rPET available? | Limited | Yes, broadly |
| Cost | $$ | $ |
| Odor retention | Low | High |
One atom over from nylon, a different material entirely.